
Print LOT number and batch designation on productsFast. Simple. Reliable.

Print LOT number and batch designation on products
You are here:
Print LOT number and batch designation
Handheld batch number printer from REINER®
Print batch number labels and batch designation on products
Mass production is part of our modern life. But what if days or weeks or months after production it is determined that an error has been made during manufacturing? Who is liable for the mistake and what must the liable party do to prevent damage to people and property?
As in previous years, we will again encounter various recall campaigns in the media 2024 in order to withdraw these defective products from circulation. The printed batch code or lot number on products plays a central role in the recall process, but also in tracing the cause of the manufacturing error.
The mobile inkjet printers from REINER® have proven themselves reliably as LOT code printers for many years and combine a multitude of advantages in one device:
• Direct printing of many batch designation types on individual parts, products, packaging, cardboard boxes
• Batch code printing on almost all smooth surfaces made of paper, cardboard, metal, plastic, glass, curved surfaces, coated labels
• Ideal for marking small parts with both fixed and serial numbers or date combined batch numbers on products
• The product batch number can be created quickly and easily on the PC using the PCset easy software supplied and transferred via USB
• The jetStamp® 990 model can be easily integrated into packaging lines as batch code printer
As in previous years, we will again encounter various recall campaigns in the media 2024 in order to withdraw these defective products from circulation. The printed batch code or lot number on products plays a central role in the recall process, but also in tracing the cause of the manufacturing error.
The mobile inkjet printers from REINER® have proven themselves reliably as LOT code printers for many years and combine a multitude of advantages in one device:
• Direct printing of many batch designation types on individual parts, products, packaging, cardboard boxes
• Batch code printing on almost all smooth surfaces made of paper, cardboard, metal, plastic, glass, curved surfaces, coated labels
• Ideal for marking small parts with both fixed and serial numbers or date combined batch numbers on products
• The product batch number can be created quickly and easily on the PC using the PCset easy software supplied and transferred via USB
• The jetStamp® 990 model can be easily integrated into packaging lines as batch code printer
What is a batch or LOT number on products?

The term “charge” comes from French and means “load”. This already indicates its origin and its importance in production and logistics. A batch is defined as the total quantity of a particular product that is produced, manufactured, or packaged under the same conditions. The producer uses the batch number to identify a delivery or production quantity that is based on the same raw material delivery, for example, or/and was produced within a work shift by the same employees, or/and was produced with the same machines, or/and another relevant production characteristic was not changed.
The batch number is printed on each item within this defined production stage that is manufactured or packaged. In this way, the manufacturer can not only have affected products removed from the supermarket shelf if necessary. In connection with a complete documentation of the production conditions he can start a tracking of the manufacturing process. In the event of complaints, the lot number helps to conduct targeted investigations into the causes of a production error.
Other common terms for this numbering are “LOT” or “Batch”. By the way, despite the frequently used capitalisation of LOT, this is not an abbreviation. It is simply the direct translation of the German word “Los”. In the case of pharmaceuticals, this designation is firmly prescribed as "Ch.-B." in accordance with §10 of the German Medicines Act [Arzneimittelgesetz] (for “batch designation” [Chargen-Bezeichnung]).
A clear legal regulation for the batch number is actually only given for medical devices. EU regulations apply to foodstuffs. For each LOT number, however, the principle applies that it must be easy to read and unambiguously identifiable. The LOT number is printed on the product packaging or directly on the product.
The batch number is printed on each item within this defined production stage that is manufactured or packaged. In this way, the manufacturer can not only have affected products removed from the supermarket shelf if necessary. In connection with a complete documentation of the production conditions he can start a tracking of the manufacturing process. In the event of complaints, the lot number helps to conduct targeted investigations into the causes of a production error.
Other common terms for this numbering are “LOT” or “Batch”. By the way, despite the frequently used capitalisation of LOT, this is not an abbreviation. It is simply the direct translation of the German word “Los”. In the case of pharmaceuticals, this designation is firmly prescribed as "Ch.-B." in accordance with §10 of the German Medicines Act [Arzneimittelgesetz] (for “batch designation” [Chargen-Bezeichnung]).
A clear legal regulation for the batch number is actually only given for medical devices. EU regulations apply to foodstuffs. For each LOT number, however, the principle applies that it must be easy to read and unambiguously identifiable. The LOT number is printed on the product packaging or directly on the product.
What is the difference between batch number and serial number and SLED?

The batch number always remains the same for the entire production lot. Only when a relevant production parameter changes, such as the addition of a new raw material delivery or the shift change of employees, is the batch number generated again and uniquely.
The serial number, on the other hand, is used to identify a single manufactured or packaged product. For example, it is incremented continuously, or provided with another unique parameter such as date-hour-minute-second. In practice, combined batch and serial numbers are often found on packaging.
The best before date (SLED) initially only says something about how long the manufacturer guarantees the perfect quality of his product. When food recalls are carried out, however, press media often say that products are recalled “with a best before date of xx to yy” printed on them. The consumer is familiar with this term and usually finds it easily on packaging. At the same time, the manufacturer can use his documentation to calculate back on which day the product was manufactured. This helps to find out what could have been the cause of the error that day.
The serial number, on the other hand, is used to identify a single manufactured or packaged product. For example, it is incremented continuously, or provided with another unique parameter such as date-hour-minute-second. In practice, combined batch and serial numbers are often found on packaging.
The best before date (SLED) initially only says something about how long the manufacturer guarantees the perfect quality of his product. When food recalls are carried out, however, press media often say that products are recalled “with a best before date of xx to yy” printed on them. The consumer is familiar with this term and usually finds it easily on packaging. At the same time, the manufacturer can use his documentation to calculate back on which day the product was manufactured. This helps to find out what could have been the cause of the error that day.

REINER® product solutions
The right device for almost every requirement.
We offer different inject printers depending on your needs and area of application.
Select the right marking device for you from our product variety:
To the handheld inkjet printer
Batch printer: Why do I have to print the LOT number in batch production?

The manufacturer of a product is always liable for his product. Now, however, it happens that a normally faultless product suddenly poses a danger to persons or property due to manufacturing errors or contaminated raw materials. Now it is up to the manufacturer to ensure that the danger is eliminated as quickly as possible.
In order to minimise the economic damage caused by the necessary recall action, it makes sense to withdraw only those products from circulation which could actually be affected by the manufacturing error. This can only be achieved by clearly tracing the end product back to its time and place of manufacture in the form of a batch number or lot number. This designation will be communicated to all dealers and customers in the recall campaign. They can then check their stored or installed products and, if necessary, return them to the manufacturer.
But batch numbers are also helpful in normal daily use, for example to obtain colours, fabrics, tiles or parquet of the same colour quality. The production of tiles on Monday may well differ slightly from the production of tiles on Tuesday. This is noticeable when laying the tiles, even if the two batches differ only slightly in appearance. This results in complaints from customers. Craftsmen therefore pay meticulous attention to always using articles of the same batch for an order.
In order to minimise the economic damage caused by the necessary recall action, it makes sense to withdraw only those products from circulation which could actually be affected by the manufacturing error. This can only be achieved by clearly tracing the end product back to its time and place of manufacture in the form of a batch number or lot number. This designation will be communicated to all dealers and customers in the recall campaign. They can then check their stored or installed products and, if necessary, return them to the manufacturer.
But batch numbers are also helpful in normal daily use, for example to obtain colours, fabrics, tiles or parquet of the same colour quality. The production of tiles on Monday may well differ slightly from the production of tiles on Tuesday. This is noticeable when laying the tiles, even if the two batches differ only slightly in appearance. This results in complaints from customers. Craftsmen therefore pay meticulous attention to always using articles of the same batch for an order.
Why is the LOT number on medical devices called “Ch.-B.” (batch designation)?

In the production of pharmaceuticals, even the smallest deviations in the dosage of active ingredients or the raw material quality of a drug can lead to serious damage to health. With § 10 of the Medicines Act, the legislator has created a binding legal requirement: Finished pharmaceuticals must, inter alia, be identified by a “batch designation”, if the medicinal product is placed on the market in batches, which is done with the abbreviation “Ch.-B.”.
For this reason, we encounter this abbreviation on all pharmaceuticals. Manufacturers are enabled to take a drug off the market in a targeted manner if a production error is detected and to investigate the causes of the defect so that it is not repeated.
For this reason, we encounter this abbreviation on all pharmaceuticals. Manufacturers are enabled to take a drug off the market in a targeted manner if a production error is detected and to investigate the causes of the defect so that it is not repeated.
How can a batch number be decoded and traced?

According to the EC Regulation, prepackaged food and many other consumer products must be labelled with a letter combination, number combination or letter/number combination that is clearly visible, legible and indelible. This combination, which each manufacturer defines for itself, is preceded by an L or LOT. The decryption usually remains a secret of the manufacturer. The only important thing for the consumer or user is actually to find the batch designation easily in order to compare it with recalls.
REINER® MarkingPartner in your area
Are you looking for an authorized dealer near you? Simply enter your country and postal code and the nearest marking partners will be displayed, like AUTOMATED MARKING INC. in the USA.
To REINER® MarkingPartner
Why are batch numbers extremely important for recalls?
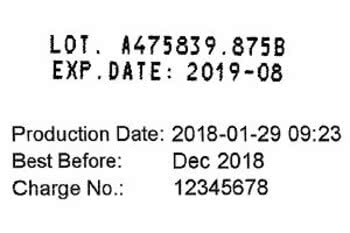
Also 2024 reports about recalls are published again and again in the media. Affected are contaminated foods, such as salmonella contaminated sausages and egg products, ski bindings, luxury cars, motorcycles, practically all industrially or handcrafted serial products. The traceability of foods and pharmaceuticals through batch numbers has often helped to protect people's health and lives.
But material assets can also be preserved if a faulty component such as a valve is replaced in good time before water damage occurs. The smaller the batch, the more important mobile and flexible marking solutions, such as our handheld batch code printing machines, become for the manufacturer.
But material assets can also be preserved if a faulty component such as a valve is replaced in good time before water damage occurs. The smaller the batch, the more important mobile and flexible marking solutions, such as our handheld batch code printing machines, become for the manufacturer.
Are labels or stickers suitable for LOT numbers?

Labels have many disadvantages. They are complex to use and require at least three work steps: 1. Printing the labels, 2. detaching it from the label carrier and 3. applying it to the object precisely. Self-adhesive labels may peel off or become illegible, especially in humid environments. The design and printing of the labels is also very complex and can deteriorate the product image.
Can a batch number be printed directly on a product?

In the case of direct marking, the batch designation is applied directly to the article or its outer packaging without an intermediate medium. Digital batch number stamps, like our Ink jet printers using suitable printer inks, are best suited for this type of industrial marking.
The mobile REINER® batch number printing machines are the clever mobile solution and complete the LOT number printing in one step:
Place the device and attach the batch designation to the goods at the push of a button – done! The robust handheld inkjet printer model REINER jetStamp® 990 does this job reliably and inexpensively. Simply generate the desired batch number with a few mouse clicks using the supplied software.
The mobile REINER® batch number printing machines are the clever mobile solution and complete the LOT number printing in one step:
Place the device and attach the batch designation to the goods at the push of a button – done! The robust handheld inkjet printer model REINER jetStamp® 990 does this job reliably and inexpensively. Simply generate the desired batch number with a few mouse clicks using the supplied software.
How can a LOT number be applied to metal, glass and plastic?

For particularly smooth surfaces, REINER® has developed a quick drying ink that remains waterproof and smear resistant immediately after printing. The innovative technology of the handheld inkjet printer jetStamp® 990 ensures the airtight sealing of the inkjet nozzles when not in use when returning them to the charging station. This prevents them from drying out and keeps these mobile marking devices functional at all times, even during long breaks.






